VIEW OUR UPCOMING VIRTUAL & IN-PERSON EVENTS
Improve Quality and Efficiency
Quality Documents and Clinical Training Management
Repeatable, documented processes are critical for GxP compliance. Your quality records set expectations for how processes and tasks should be conducted. Trial Interactive provides a central, controlled workspace to add quality record templates and track them through draft, review, and approval before making them effective. Seamlessly send your records to the GlobalLearn LMS to train teams on clinical trial records to enforce quality compliance.
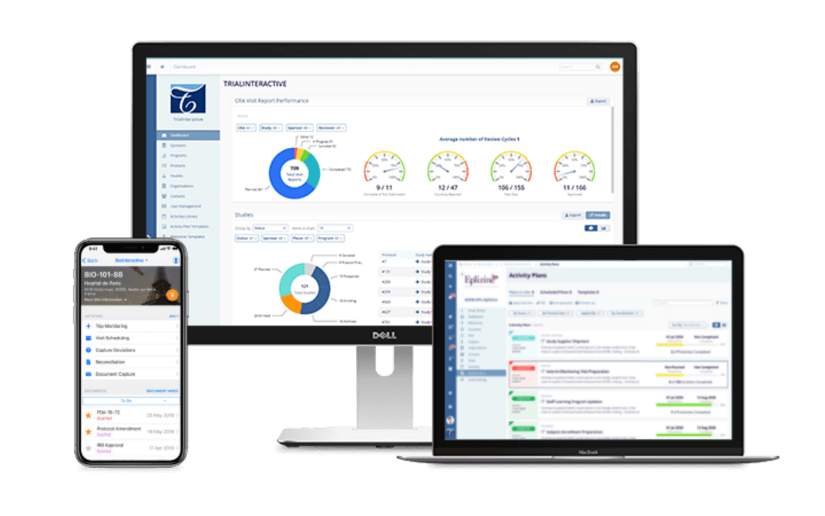
Get the Benefits of TI Quality Records
-
Simplify QA Record Processes
-
Ensure GxP and GMP Compliance
-
Automate Quality Record Processes
-
Improve Collaboration
-
Maintain Complete Audit Trail and Record History
-
Manage Record Versioning
-
Control Access to Clinical Trial Records with Edit and Read-Only Permissions
-
Reduce Costs with Centralized Records and Training Management
-
Monitor Record Processes with Real-Time Oversight
-
Track Training Progress, Compliance, and Completion
-
Certify Team Members on Quality Processes through Training Management
-
Publish Quality Records Directly to the eTMF
-
Publish Training Certifications to the eTMF
Ensure Compliance
Quality assurance teams can manage standard operating procedures, policies, and any other records from approved templates as required by the quality manual. Author, edit, approve, and manage quality records.
Simplify quality knowledge management by connecting quality document processes to your training management.
-
Draft from Template, Edit and Collaborate on Document
-
Approve with eSignature
-
Share Approved Document to Training Management
-
Document Becomes Effective

