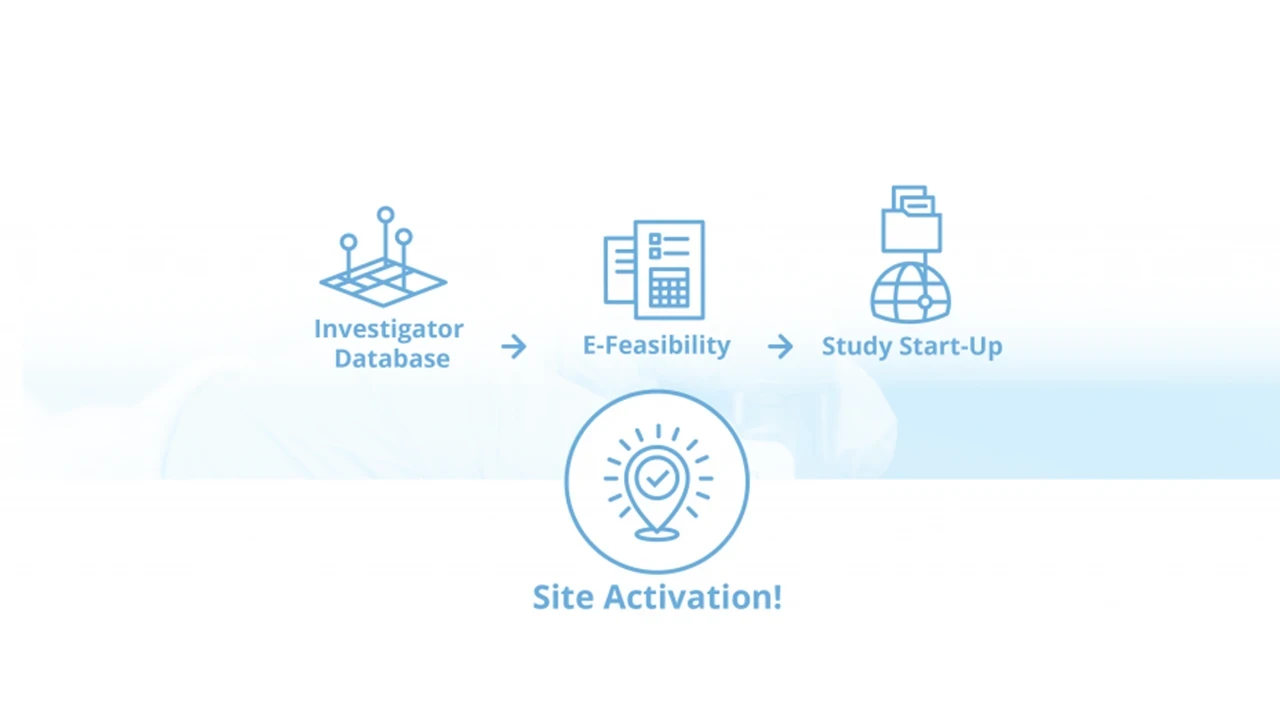
Investigator Database Highlights
- Search an Exhaustive Collection of Public Data
- Leverage Your Historical Site/Investigator Data
- Search & Filter Sites and Investigators
- Save Site/Investigator Collections
- Identify Sites Through Study Start-Up
- Speed SFQs & Enhance Site/Investigator Data
- Multilingual Translation Support
- 24/7 Help Desk (Email/Phone Support)